Evolution of enzymes involved in lipid biosynthesis
Insect species use pheromones composed of different secondary metabolites for mate finding and mating. Pheromones are specific mixtures of saturated and unsaturated fatty acid (FA)-derived alcohols, aldehydes, esters, hydrocarborns, and epoxides and contribute to evolutionary success. Subtle changes in pheromone composition can represent the origin of new species evolution.
The overall goal of this work is to uncover molecular mechanisms for the evolution of pheromone components. We focus on classes of enzymes involved in fatty acyl derivative biosynthesis, such as membrane fatty acyl desaturases (mFADs, EC 1.14.19.5) and fatty acyl reductases (FARs, EC 1.2.1.84). mFADs introduce double bonds into FA hydrocarbon chains, and FARs reduce fatty-acyl CoA to fatty alcohols. As model species, we use the tobacco hornworm moth, Manduca sexta (Lepidoptera), and the bumblebees Bombus terrestris, Bombus lucorum, and Bombus lapidarius.
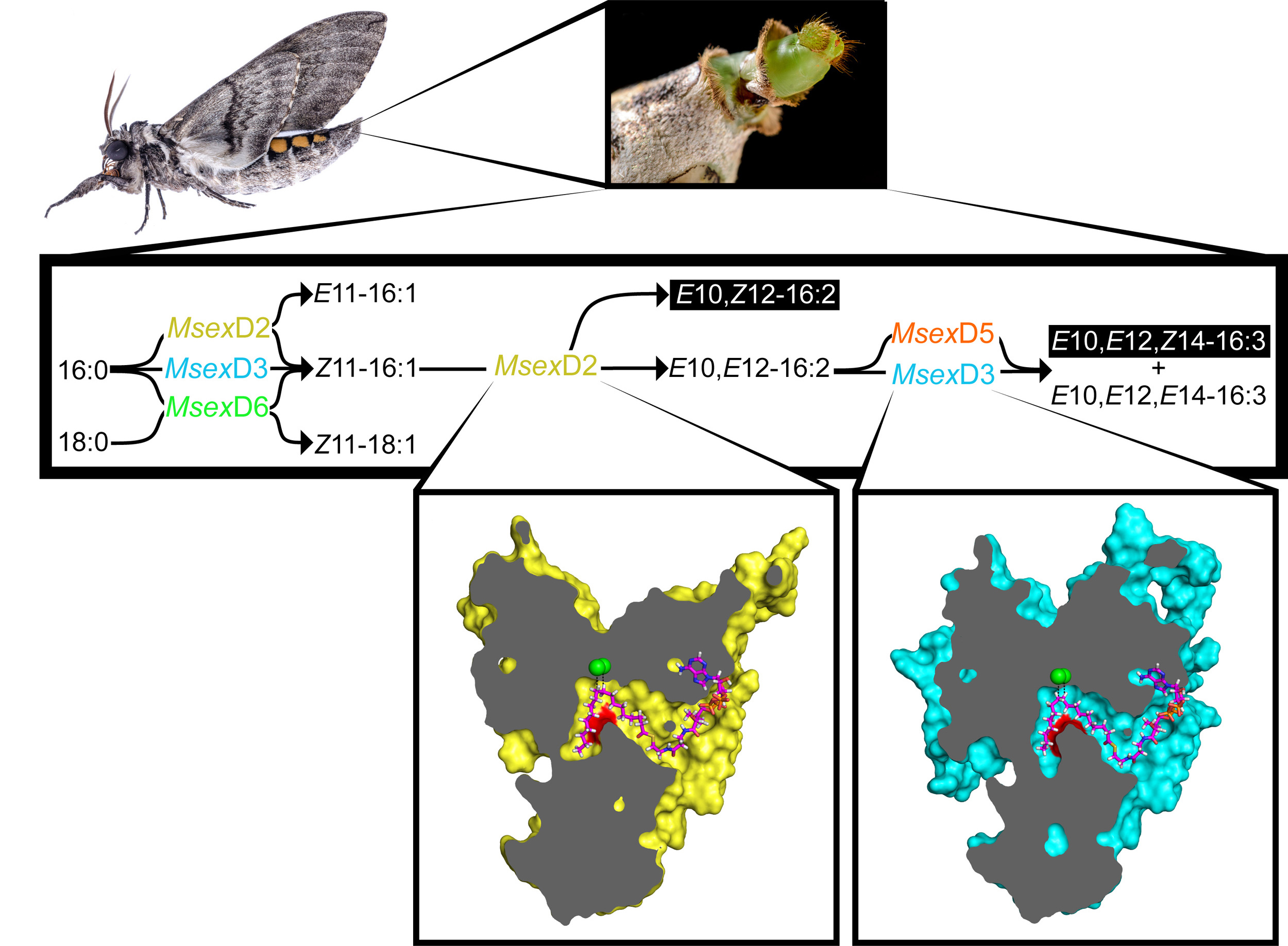
We have found that a single amino acid substitution in the kink of the mFAD substrate binding tunnel can lead to synthesis of novel unsaturated FA precursors. Currently, our project is focused on confirming this mechanism of FAD specificity determination and understanding the evolution of new pheromone components and new insect species.
Novel results obtained from analysis of FAR gene expression profiles in pheromone glands of three bumblebee species indicate that these insects possess a separate class of FARs. We are working to characterize these enzymes and determine the structural motifs critical for their functions.
Our manuscript published in PNAS in 2015 inspired René Levinski’s theatre performance “Press Space to Continue,” which was on the program of the New Stage of the National Theatre in Prague and the City Theatre of Brno.
Our publications (2010-)
- Tupec M., Culka M., Machara A., Macháček S., Bím D., Svatoš A., Rulíšek L., Pichová I.: Understanding desaturation/hydroxylation activity of castor stearoyl Δ9-Desaturase through rational mutagenesis. Comput. Struct. Biotechnol. J. 2022, 20:1378-1388.
- Buček A., Vazdar M., Tupec M., Svatoš A., Pichová I.: Desaturase specificity is controlled by the physicochemical properties of a single amino acid residue in the substrate binding tunnel. Comput. Struct. Biotechnol. J. 2020, 18:1202-1209.
- Tupec M., Buček A., Janoušek V., Vogel H., Prchalová D., Kindl J., Pavlíčková T., Wenzelová P., Jahn U., Valterová I., Pichová I.: Expansion of the fatty acyl reductase gene family shaped pheromone communication in Hymenoptera. eLife. 2019, 8:e39231.
- Tupec M., Buček A., Valterová I., Pichová I.: Biotechnological potential of insect fatty acid-modifying enzymes. Z. Naturforsch. C. 2017, 72(9-10):387-403.
- Buček A., Brabcová J., Vogel H., Prchalová D., Kindl J., Valterová I., Pichová I.: Exploring complex pheromone biosynthetic processes in the bumblebee male labial gland by RNA sequencing. Insect Mol. Biol. 2016, 25:295-314.
- Prchalová D., Buček A., Brabcová J., Žáček P., Kindl J., Valterová I., Pichová I.: Regulation of Isoprenoid Pheromone Biosynthesis in Bumblebee Males. ChemBioChem. 2016, 17:260.
- Buček A., Matoušková P., Vogel H., Šebesta P., Jahn U., Weißflog J., Svatoš A., Pichová I.: Evolution of moth sex pheromone composition by a single amino acid substitution in a fatty acid desaturase. Proc. Natl. Acad. Sci. U.S.A. 2015, 112(41):12586-12591.
- Buček A., Vogel H., Matoušková P., Prchalová D., Žáček P., Vrkoslav V., Šebesta P., Svatoš A., Jahn U., Valterová I., Pichová I.: The role of desaturases in the biosynthesis of marking pheromones in bumblebee males. Insect Biochem. Mol. Biol. 2013, 43(8):724-731.
- Žáček P., Prchalová‐Horňáková D., Tykva R., Kindl J., Vogel H., Svatoš A., Pichová I., Valterová I.: De Novo Biosynthesis of Sexual Pheromone in the Labial Gland of Bumblebee Males. ChemBioChem. 2013, 14:361-371.
- Horňáková D., Matoušková P., Kindl J., Valterová I., Pichová I.: Selection of reference genes for real-time polymerase chain reaction analysis in tissues from Bombus terrestris and Bombus lucorum of different ages. Anal. Biochem. 2010, 397(1):118-120.
Collaboration
- Robert Hanus (IOCB)
- Lubomír Rulíšek (IOCB)
- Pavel Majer (IOCB)
People
Michal Tupec, Stanislav Macháček, Martina Halmová

